Physics Was Never so Easy!
Heat
Heat is the total amount of energy content of a body. In daily life we
usually confuse heat with temperature, but temperature is very different from
heat as it is the average kinetic energy of a body.
Heat's SI unit is Joules or J
Temperatures SI unit is Kelvin or K
usually confuse heat with temperature, but temperature is very different from
heat as it is the average kinetic energy of a body.
Heat's SI unit is Joules or J
Temperatures SI unit is Kelvin or K
To calculate the amount of energy needed to raise a body's temperature by a
certain formula can be used
Q=mc(ˆt)
where Q is the amount of energy;
m is mass of the body;
c is the specific heat capacity of the body;
and ˆt is the change in temperature that need to be brought about or t2-t1 (temp. after heating minus temp. before heating.)
certain formula can be used
Q=mc(ˆt)
where Q is the amount of energy;
m is mass of the body;
c is the specific heat capacity of the body;
and ˆt is the change in temperature that need to be brought about or t2-t1 (temp. after heating minus temp. before heating.)
Transfer of Heat
The Transfer of heat can take place by 3 methods Conduction,Convection and
Radiation.
Radiation.
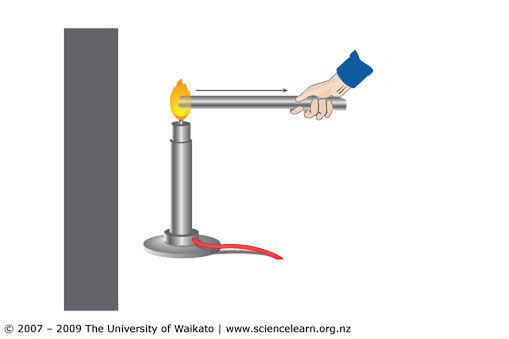
Conduction
Conduction takes place only in solids because in conduction the particles dont
move but directly transfer the heat to the other particles (the inter-molecular
spaces in solids are very less)
Conductors are certain substances which are efficient in the transfer of heat or
electricity (usually metals).
move but directly transfer the heat to the other particles (the inter-molecular
spaces in solids are very less)
Conductors are certain substances which are efficient in the transfer of heat or
electricity (usually metals).
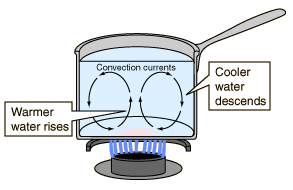
Convection
Convection takes place only in liquids and gasses. In this mode of transfer of
heat the particles actually move and hit each other and transfer the energy
(this is able to take place as the inter-molecular spaces in liquids and gasses
are much greater than in solids)
In convection, convection currents are created, in Liquids when the liquid is
heated the heated part of the liquid rises and the cooler part of the liquid
descends to be heated, htis process is repeated again and again. The same
principle applies to gasses.
heat the particles actually move and hit each other and transfer the energy
(this is able to take place as the inter-molecular spaces in liquids and gasses
are much greater than in solids)
In convection, convection currents are created, in Liquids when the liquid is
heated the heated part of the liquid rises and the cooler part of the liquid
descends to be heated, htis process is repeated again and again. The same
principle applies to gasses.
Radiation
Radiation is the only mode of transfer of heat which does not need a medium.
We get the suns rays via radiation. We know this as space is a vacuum and
yet we feel hot on earth.
We get the suns rays via radiation. We know this as space is a vacuum and
yet we feel hot on earth.